

This BCS sub-classification is particularly important for in vivo predictive dissolution methodology development due to the complex and variable in vivo environment in the gastrointestinal tract, with its changing pH, buffer capacity, luminal volume, surfactant luminal conditions, permeability profile along the gastrointestinal tract and variable transit and fasted and fed states. It is well known that the, p Ka physical property of a drug (API) has a significant impact on the aqueous solubility dissolution of drug from the drug product both in vitro and in vivo for BCS Class II and IV acids and bases, and is the basis, we propose for a sub-classification extension of the original BCS classification.
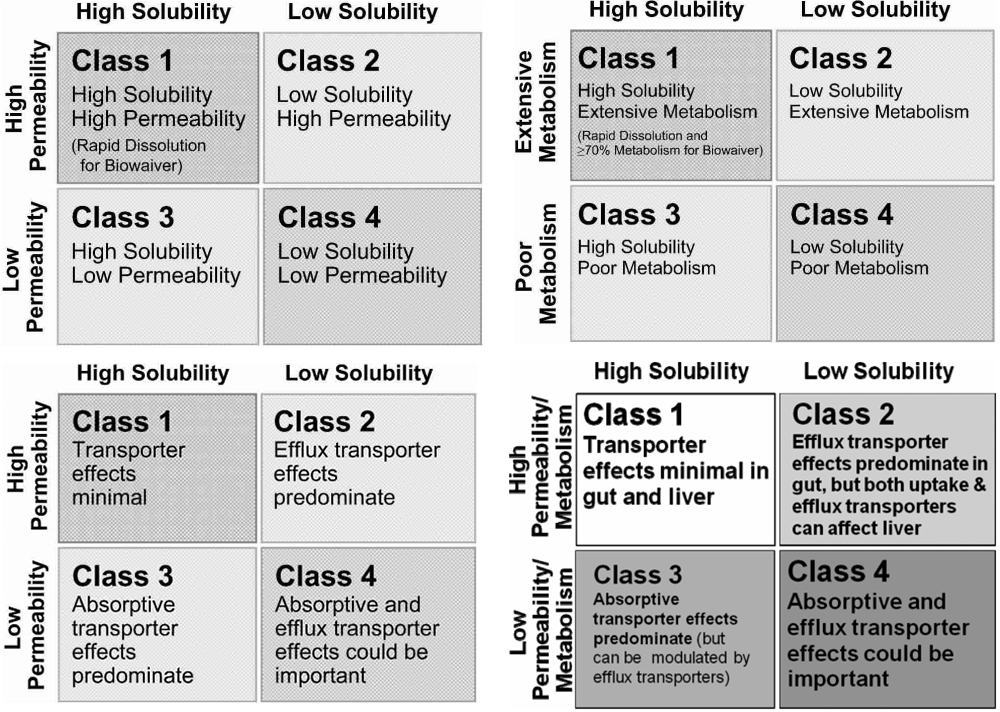
Sub-classification for Classes I and III (high solubility drugs as currently defined) is generally not needed except perhaps in border line solubility cases.

In this manuscript we propose a simple extension of the BCS classes to include subspecification of acid (a), base (b) and neutral (c) for classes II and IV. Nevertheless, underneath the simplicity of BCS are many detailed complexities, both in vitro and in vivo which must be evaluated and investigated for any given drug and drug product. The mechanistic base of the BCS approach has, no doubt, contributed to its wide spread acceptance and utility. The classification scheme captures the two most significant factors influencing oral drug absorption solubility and intestinal permeability and it has proven to be a very useful and a widely accepted starting point for drug product development and drug product regulation. The Biopharmaceutics Classification System (BCS) has found widespread utility in drug discovery, product development and drug product regulatory sciences.

 RSS Feed
RSS Feed
